 Density depends on the mass of an individual molecule and the number of such molecules that occupy a unit of volume. Except in certain special cases, such as the flow of a compressible gas (in which the density is not constant) or a liquid under a very high shear rate (in which viscous dissipation can cause significant internal heating), or situations involving exothermic or endothermic reactions, we shall ignore any variation of physical properties with pressure and temperature.ĭensity. 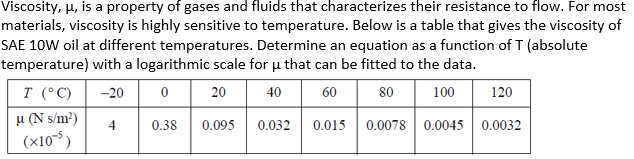
Typical processes often run almost isothermally, and in these cases the effect of temperature can be ignored. The density of gases depends almost directly on the absolute pressure for most other cases, the effect of pressure on physical properties can be disregarded. For liquids, viscosity also depends strongly on the temperature for gases, viscosity is approximately proportional to the square root of the absolute temperature. The physical properties depend primarily on the particular fluid. Each of these will be defined and viewed briefly in terms of molecular concepts, and their dimensions will be examined in terms of mass, length, and time (M, L, and T). 
There are three physical properties of fluids that are particularly important: density, viscosity, and surface tension. 
Learn More Buy 1.4 Physical Properties-Density, Viscosity, and Surface Tension Fluid Mechanics for Chemical Engineers: with Microfluidics, CFD, and COMSOL Multiphysics 5, 3rd Edition 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
